Software for integrating ODEs
Background
Numerically integrating ODE's is something you should have mastered in your pre-requisite 3E4 course. If you are not familiar with this topic, it is your responsibility to quickly catch up, because we will use this intensively for the rest of the course.
Here is a tutorial a wrote a few years ago when I taught 3E4 in 2010. The tutorial below is similar, but uses a reactor design example.
Example
The example we will work with is a common modelling reaction: a liquid-based stirred tank reactor, with (initially) constant physical properties, a second order chemical reaction where species A is converted to B according to
where
In the code below we will integrate the ODE from
MATLAB code
In a file called tank.m:
function d_depnt__d_indep = tank(indep, depnt)
% Dynamic balance for a CSTR
%
% C_A = y(1) = the concentration of A in the tank, mol/L
%
% Returns dy/dt = F/V*(C_{A,in} - C_A) - k*C_A^2
% indep: the independent ODE variable, such as time or length or the reactor
% depnt: a VECTOR of dependent variables
%
% Returns:
%
% d(depnt)
% ---------- = a vector of ODEs
% d(indep)
% Assign some variables for convenience of notation: one row per DEPENDENT variable
CA = depnt(1);
% Constant and other equations
F = 20.1; % L/min
CA_in = 2.5; % mol/L
V = 100; % L
k = 0.150; % L/(min.mol)
% The output from the ODE function must be a COLUMN vector, with n rows
% n = how many ODE's in this system?
n = numel(depnt); d_depnt__d_indep = zeros(n,1);
% Specify every element in the vector below: 1, 2, ... n
d_depnt__d_indep(1) = F/V*(CA_in - CA) - k*CA^2;
In a separate file (any name), for example: ode_driver.m, which will "drive" the ODE solver:
% Integrate the ODE
% -----------------
% Set the time range:
t_start = 0;
t_final = 10.0;
% Set the initial condition(s):
CA_t_zero = 0.5;
% Integrate the ODE(s):
[t, y] = ode45(@tank, [t_start, t_final], [CA_t_zero]);
% Plot the results:
clf;
plot(t, y)
grid('on')
xlabel('Time [minutes]')
ylabel('Concentration of A [mol/L]')
A plot from this code shows the system stabilizing after about 9 minutes.
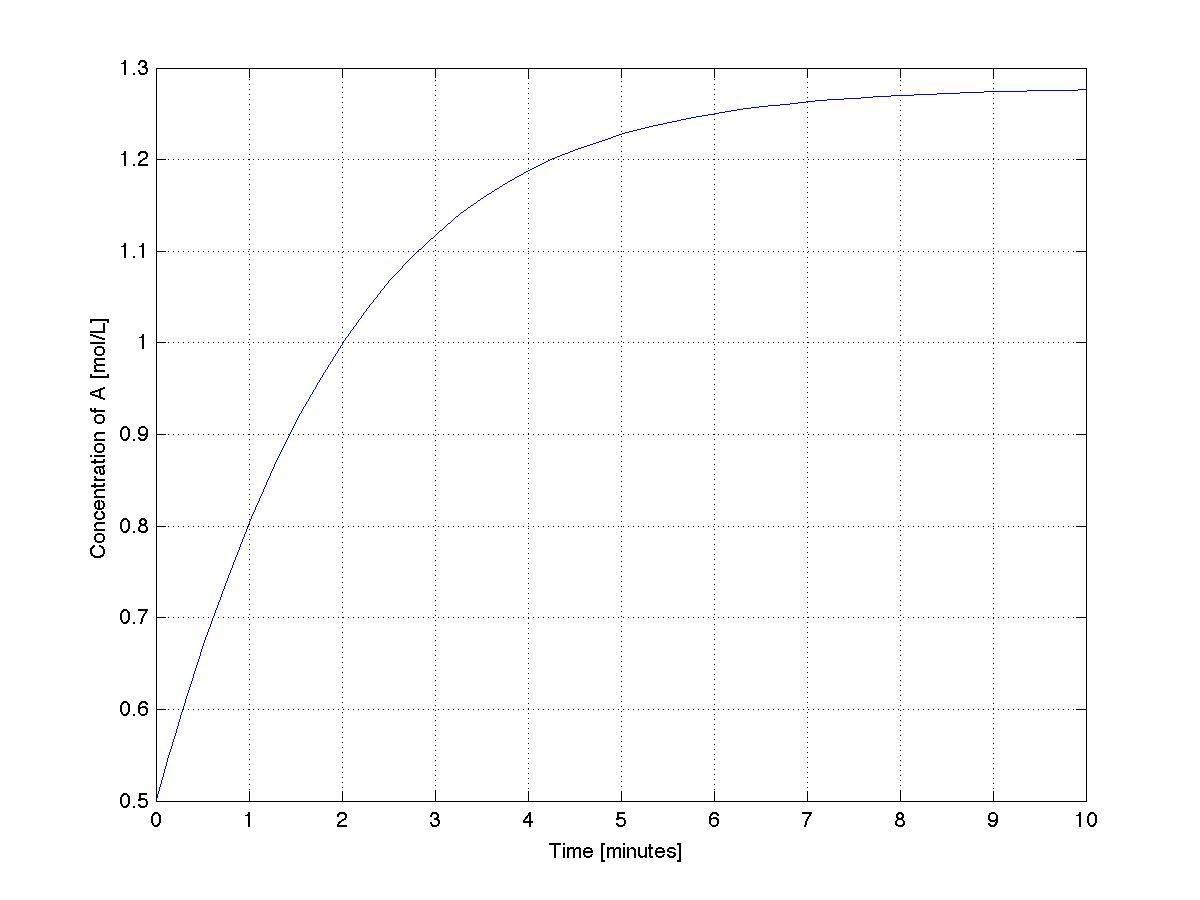
Let's take a look at the MATLAB output:
>> t(1:10)
ans =
0
0.0689
0.1378
0.2067
0.2757
0.5257
0.7757
1.0257
1.2757
1.5257
>> y(1:10)
ans =
0.5000
0.5248
0.5490
0.5726
0.5956
0.6742
0.7452
0.8090
0.8662
0.9171
MATLAB places the points at unequal step sizes of time. This is what their documentation has to say:
- The MATLAB ODE solvers utilize these methods by taking a step, estimating the error at this step, checking to see if the value is greater than or less than the tolerance, and altering the step size accordingly. These integration methods do not lend themselves to a fixed step size. Using an algorithm that uses a fixed step size is dangerous since you can miss points where your signal frequency is greater than the solver frequency. Using a variable step ensures that a large step size is used for low frequencies and a small step size is used for high frequencies. The ODE solvers within MATLAB are optimized for a variable step, run faster with a variable step size, and clearly the results are more accurate.
Example with coupled ODEs
We will now expand our example to a system of two coupled ODEs. Still in the same reactor we are now considering the rate constant to be a function of temperature:
where
In the code below we will integrate the ODE from
The following code appears in an m-file called: tank_coupled.m
function d_depnt__d_indep = tank_coupled(indep, depnt)
% Dynamic balance for a CSTR
% C_A = y(1) = the concentration of A in the tank, [mol/L]
% T = y(2) = the tank temperature, [K]
%
% Returns dy/dt = [F/V*(C_{A,in} - C_A) - k*C_A^2 ]
% [F/V*(T_in - T) - k*C_A^2*HR/(rho*Cp) ]
%
% indep: the independent ODE variable, such as time or length or the reactor
% depnt: a VECTOR of dependent variables
%
% Returns:
%
% d(depnt)
% ---------- = a vector of ODEs
% d(indep)
% Assign some variables for convenience of notation: one row per DEPENDENT variable
CA = depnt(1);
T = depnt(2);
% Constants
F = 20.1; % L/min
CA_in = 2.5; % mol/L
V = 100.0; % L
k0 = 0.15; % L/(mol.min)
Ea = 5000; % J/mol
R = 8.314; % J/(mol.K)
Hr = -590; % J/mol
T_in = 288; % K
rho = 1.050; % kg/L
% Algebraic equations
k = k0 * exp(-Ea/(R*T)); % L/(mol.min)
Cp = 4.184 - 0.002*(T-273); % J/(kg.K)
% Output from this ODE function must be a COLUMN vector, with n rows
% n = how many ODEs in this system?
n = numel(depnt); d_depnt__d_indep = zeros(n,1);
d_depnt__d_indep(1) = F/V*(CA_in - CA) - k*CA^2;
d_depnt__d_indep(2) = F/V*(T_in - T) - (Hr*k*CA^2)/(rho*Cp);
In a separate file (any name), for example: ode_driver.m, which will "drive" the ODE solver:
% Integrate the ODE
% -----------------
% Set the time range:
t_start = 0;
t_final = 45.0;
% Set the initial condition(s):
CA_t_zero = 0.5;
T_t_zero = 295;
% Integrate the ODE(s):
[t, y] = ode45(@tank_coupled, [t_start, t_final], ...
[CA_t_zero, T_t_zero]);
% Plot the results:
clf;
subplot(2, 1, 1)
plot(t, y(:,1))
xlabel('Time [minutes]')
ylabel('Concentration of A [mol/L]')
grid('on')
subplot(2, 1, 2)
plot(t, y(:,2), 'r')
xlabel('Time [minutes]')
ylabel('Temperature in tank [K]')
grid('on')
print('-dpng', 'coupled-ode-MATLAB.png')
The trajectories produced by the above code are shown below. Do they make engineering sense?
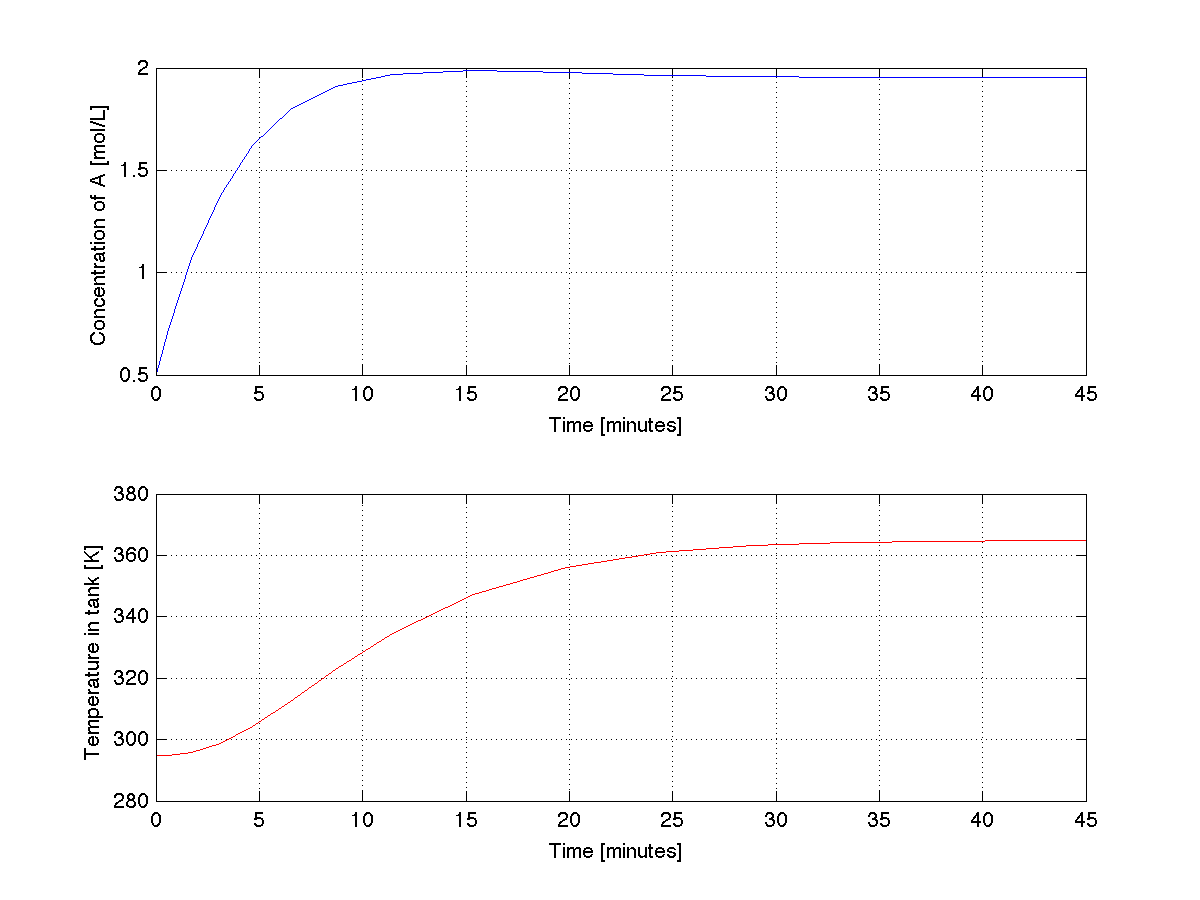
- Important notes
- Each trajectory (ODE) must have an initial condition.
- The function that specifies the ODE must return a column vector with
- Algebraic equations may be included in the ODE function. For example:
- If the inlet flow varied over time according to
- Numerical integration allows for discontinuities. For example, a step input at
if t < 50
F = 20.1
else
F = 25.1
end